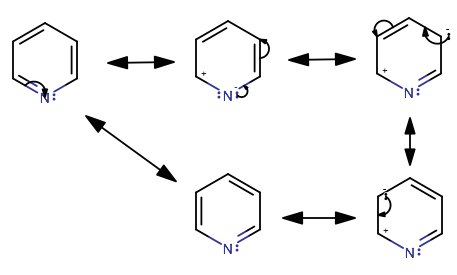
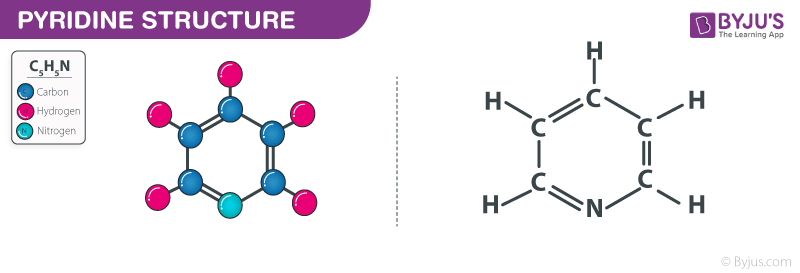
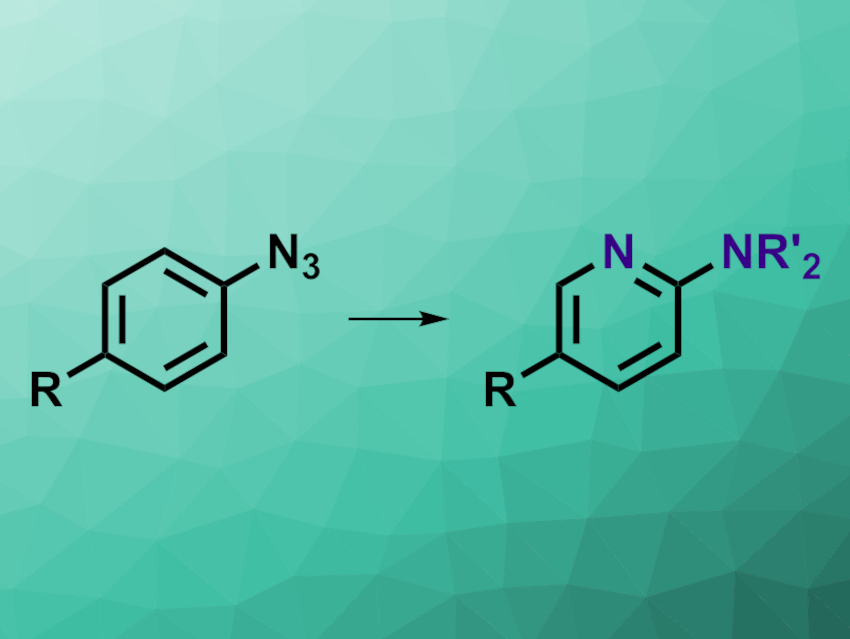
Sequential Ring-Opening and Ring-Closing Reactions for Converting para-Substituted Pyridines into meta-Substituted Anilines | Organic Letters
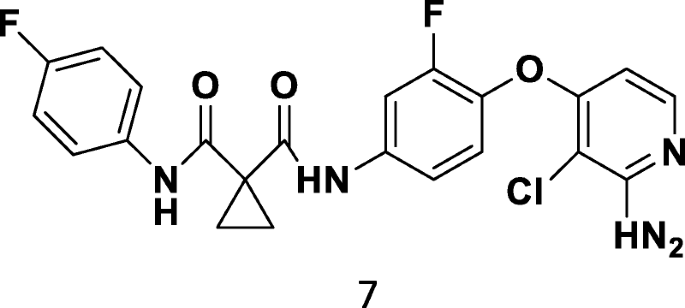
Medicinal attributes of pyridine scaffold as anticancer targeting agents | Future Journal of Pharmaceutical Sciences | Full Text

Pyridine Ring Stock Illustrations – 7 Pyridine Ring Stock Illustrations, Vectors & Clipart - Dreamstime

Non-enzymatic pyridine ring formation in the biosynthesis of the rubrolone tropolone alkaloids | Nature Communications