
Development of an Efficient Procedure for Indole Ring Synthesis from 2-Ethynylaniline Derivatives Catalyzed by Cu(II) Salts and Its Application to Natural Product Synthesis | The Journal of Organic Chemistry

Synthetic route and atom numbering of indole enaminoketones 1c, 2c and... | Download Scientific Diagram
The structural formulae of indole-3-acetic acid, indole-3-ethanol and... | Download Scientific Diagram
![Pharmaceuticals | Free Full-Text | Synthesis and Biological Activities of Pyrazino[1,2-a]indole and Pyrazino[1,2-a]indol-1-one Derivatives Pharmaceuticals | Free Full-Text | Synthesis and Biological Activities of Pyrazino[1,2-a]indole and Pyrazino[1,2-a]indol-1-one Derivatives](https://pub.mdpi-res.com/pharmaceuticals/pharmaceuticals-14-00779/article_deploy/html/images/pharmaceuticals-14-00779-g001.png?1628414957)
Pharmaceuticals | Free Full-Text | Synthesis and Biological Activities of Pyrazino[1,2-a]indole and Pyrazino[1,2-a]indol-1-one Derivatives

Intramolecular Fischer Indole Synthesis for the Direct Synthesis of 3,4-Fused Tricyclic Indole and Application to the Total Synthesis of (−)-Aurantioclavine | Organic Letters
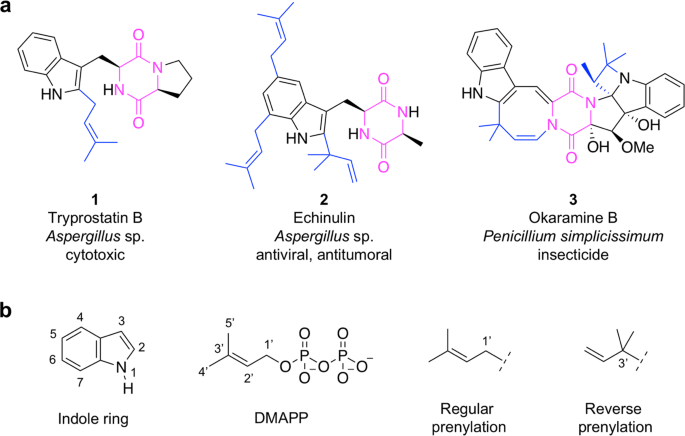
Reprogramming Escherichia coli for the production of prenylated indole diketopiperazine alkaloids | Scientific Reports



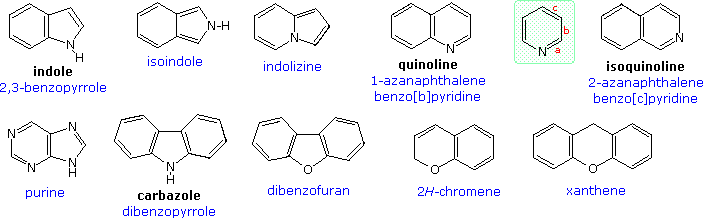

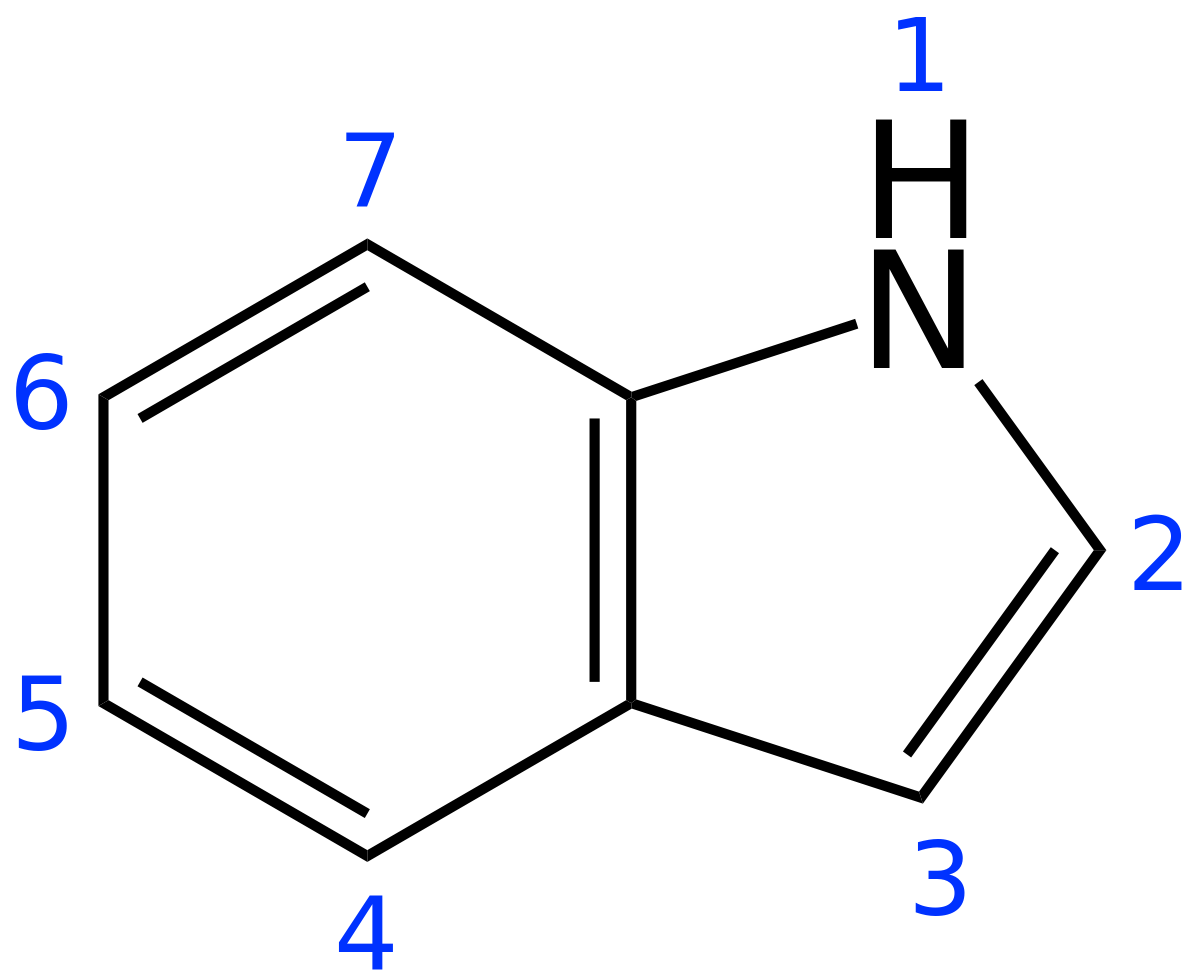

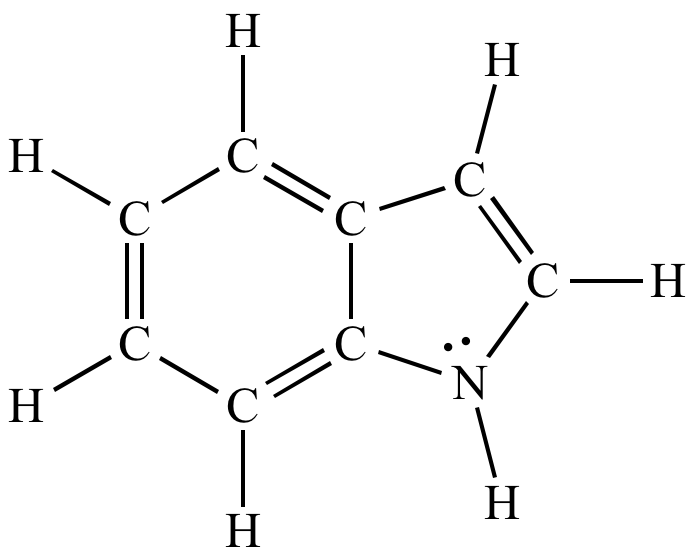
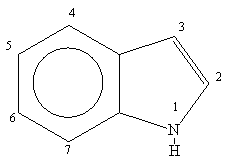
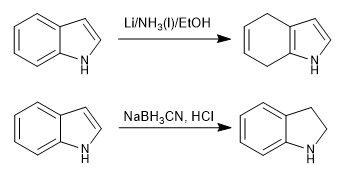

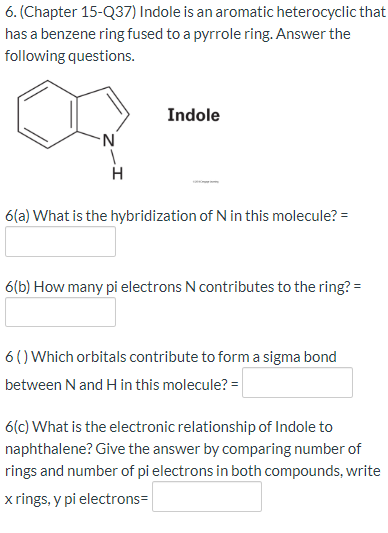

![Indole] Indole]](https://www.degruyter.com/document/doi/00.0000/IUPAC.iupac.compound.798/asset/images/798.png)