Darzalex (Daratumumab): First Anti-CD38 Monoclonal Antibody Approved for Patients with Relapsed Multiple Myeloma
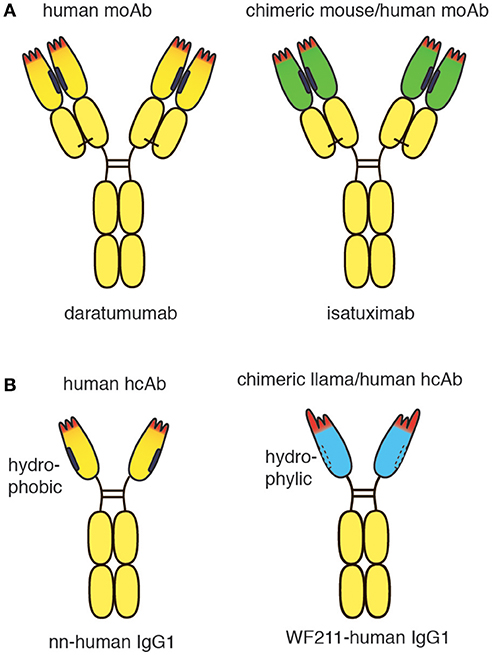
Frontiers | Perspectives for the Development of CD38-Specific Heavy Chain Antibodies as Therapeutics for Multiple Myeloma
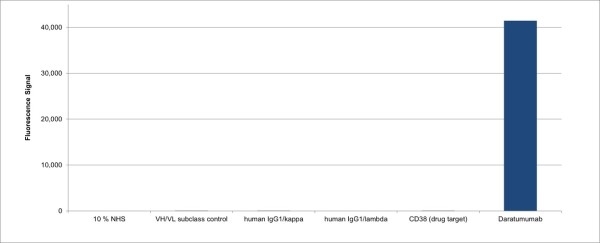
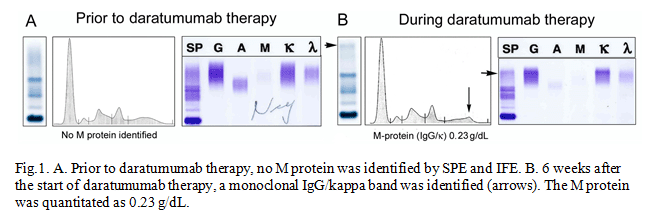
Diagnostics | Free Full-Text | Complete Depletion of Daratumumab Interference in Serum Samples from Plasma Cell Myeloma Patients Improves the Detection of Endogenous M-Proteins in a Preliminary Study
Diagnostics | Free Full-Text | Complete Depletion of Daratumumab Interference in Serum Samples from Plasma Cell Myeloma Patients Improves the Detection of Endogenous M-Proteins in a Preliminary Study

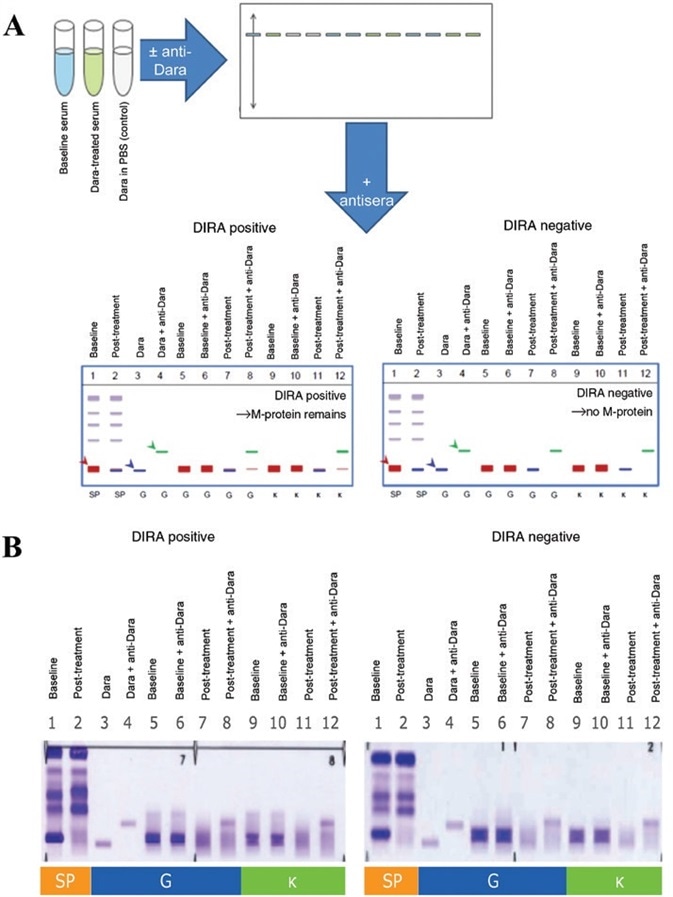
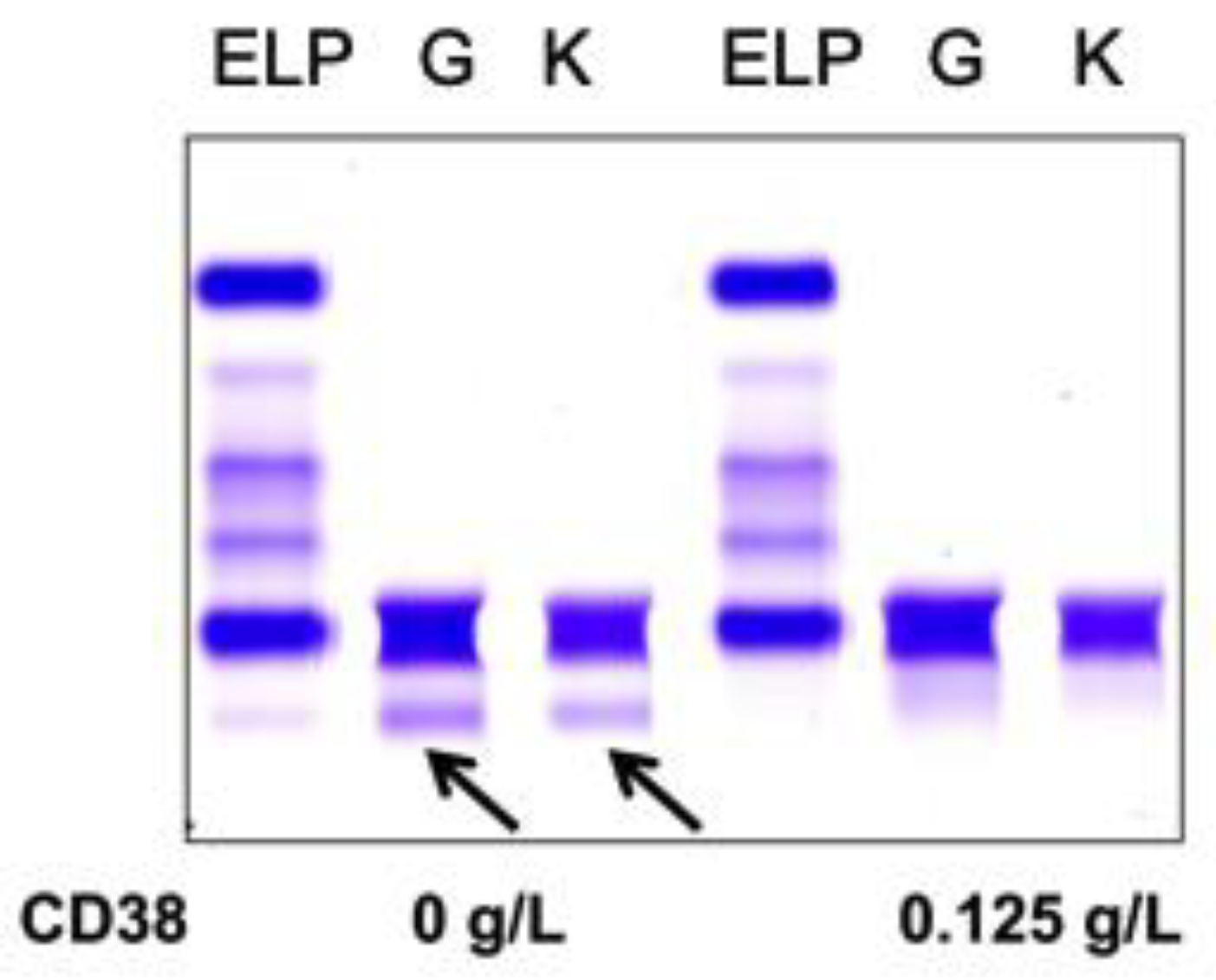
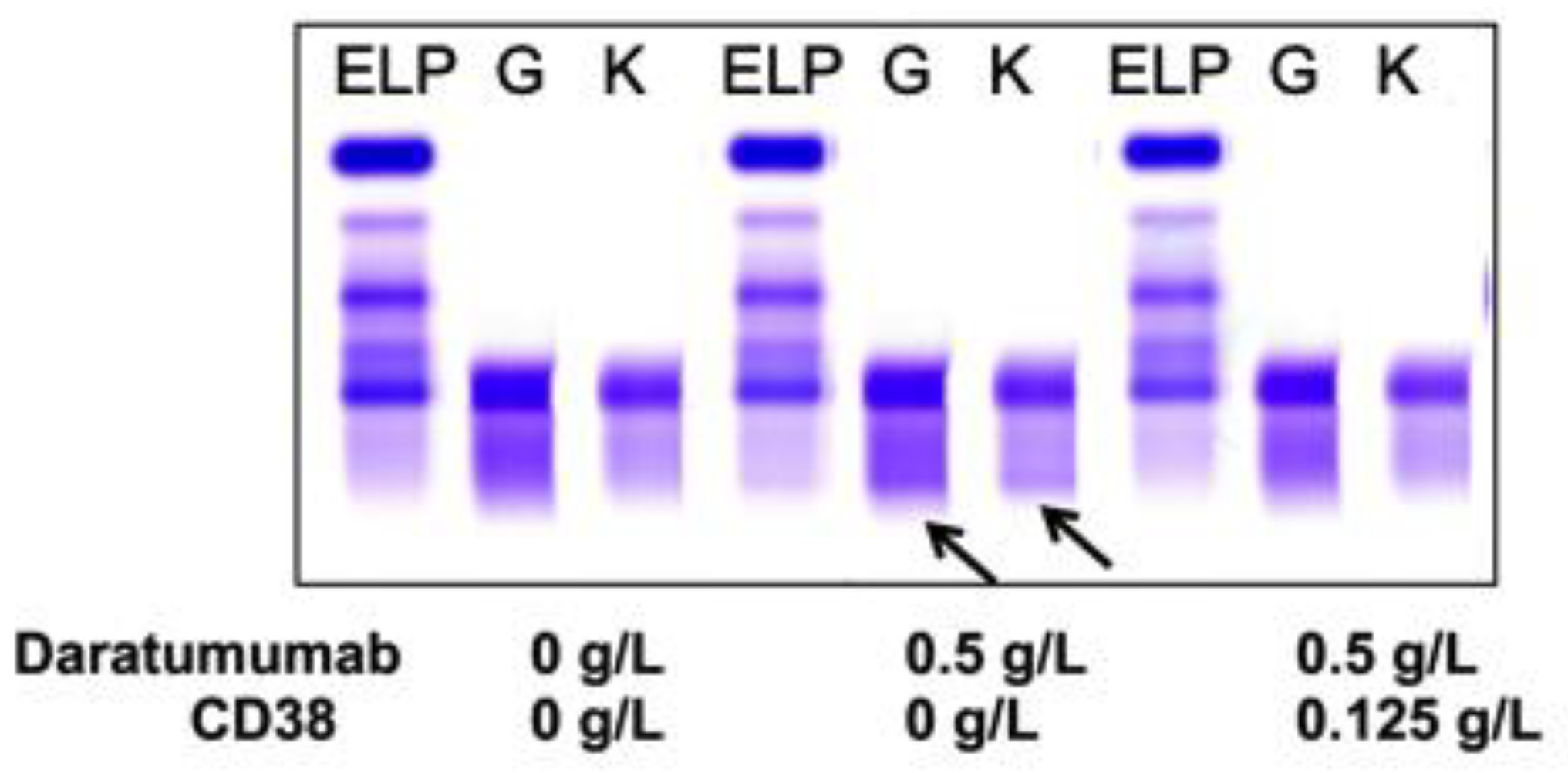
Figure 1 from Interference of daratumumab in monitoring multiple myeloma patients using serum immunofixation electrophoresis can be abrogated using the daratumumab IFE reflex assay (DIRA) | Semantic Scholar

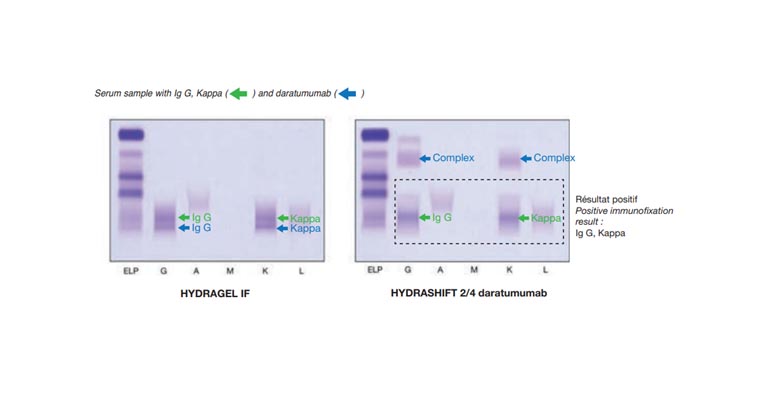
Monitoring the M-protein of multiple myeloma patients treated with a combination of monoclonal antibodies: the laboratory solution to eliminate interference

Monitoring multiple myeloma patients treated with daratumumab: teasing out monoclonal antibody interference

Investigation into the interference of the monoclonal antibody daratumumab on the free light chain assay. | Semantic Scholar
DARZALEX® (daratumumab) Approved by U.S. FDA in Combination with Two Standard of Care Regimens for the Treatment of Patients with Multiple Myeloma Who Have Received At Least One Prior Therapy

Investigation into the interference of the monoclonal antibody daratumumab on the free light chain assay - ScienceDirect

Implications for the monitoring of patients with multiple myeloma undergoing treatment with the anti-CD38 monoclonal daratumumab - Sally Thirkettle, Joanne Russell, Sarah Wilson, Tasneem Ganijee, Samar Kulkarni, Jim Cavet, Phillip J Monaghan,

Current use of monoclonal antibodies in the treatment of multiple myeloma - Varga - 2018 - British Journal of Haematology - Wiley Online Library
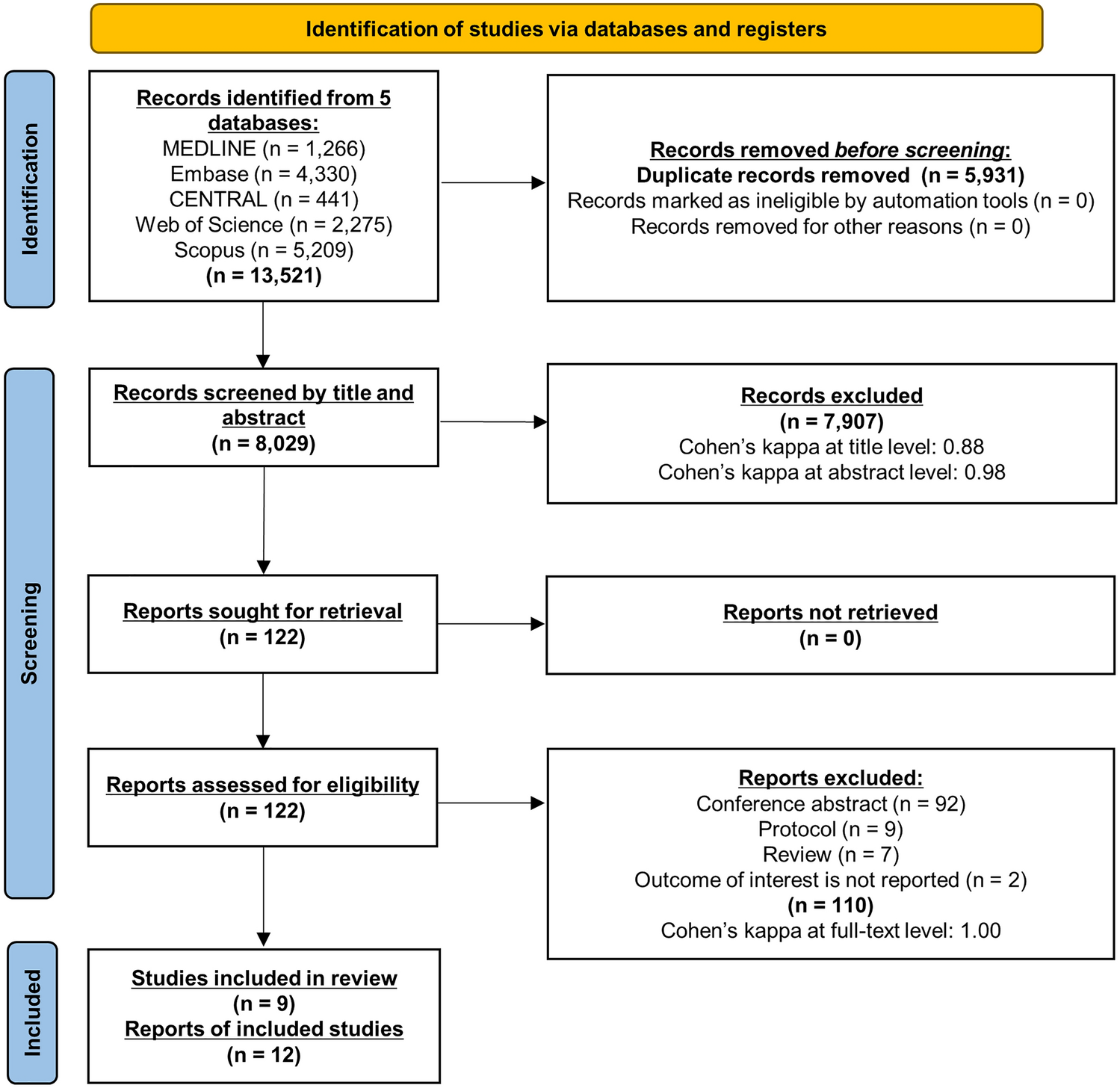
Addition of daratumumab to multiple myeloma backbone regimens significantly improves clinical outcomes: a systematic review and meta-analysis of randomised controlled trials | Scientific Reports
U.S. Food and Drug Administration Approves DARZALEX FASPRO™ (daratumumab and hyaluronidase-fihj), a New Subcutaneous Formulation of Daratumumab in the Treatment of Patients with Multiple Myeloma