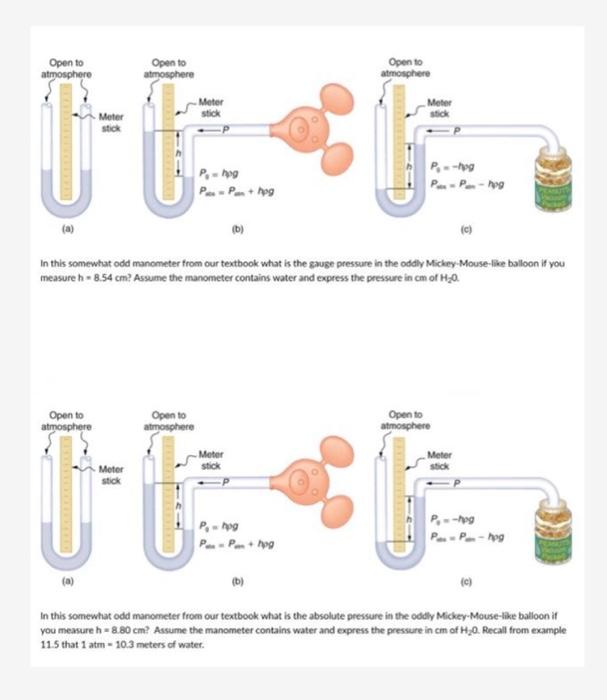
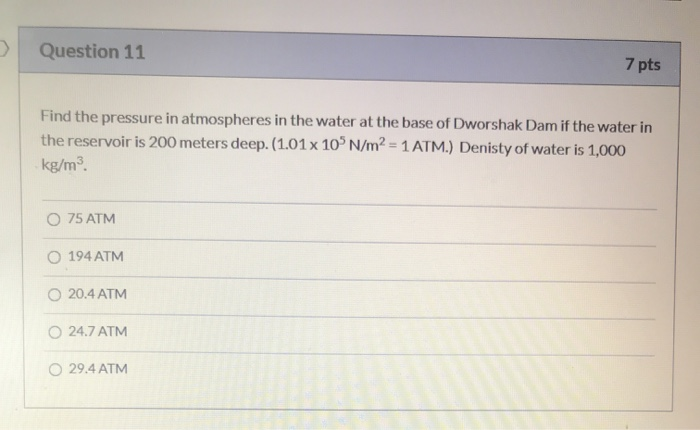
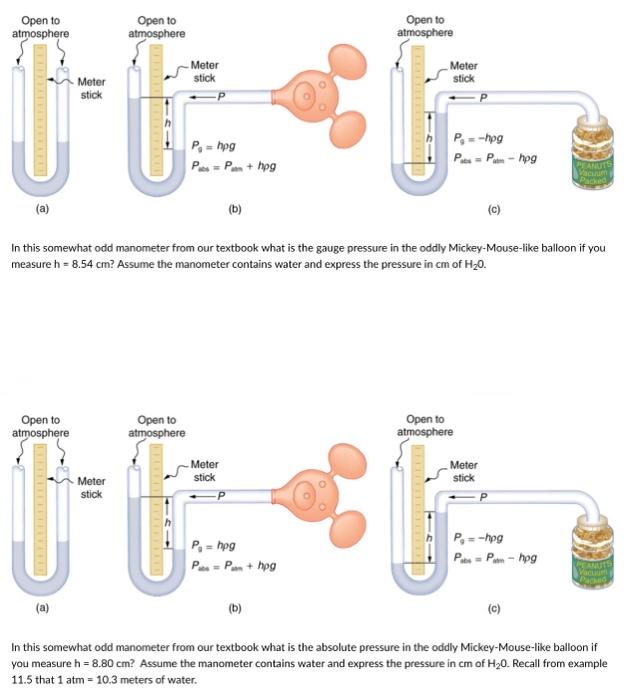
SOLVED: The pressure 35.( mcters under water is 445 kPa. Conversion fectors 1kPa 9.869 atm atm 780 mmHg Whal is this pressure in atm? pressure: What this pressure in mmHg? mmdg pressurc:
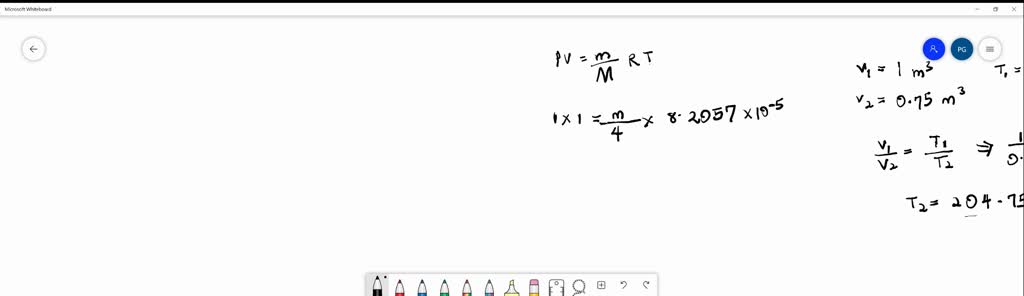
SOLVED: A cubic meter of helium originally at 0-degrees C and 1-atm pressure is cooled at constant pressure until the volume is 0.75 𝑚^3.. How much heat in kcal is removed? CvHelium =